|
There they can be collected and analyzed in terms of their numbers at specific kinetic energies, typically with electrostatic lenses and hemispherical analyzers. However, a certain fraction (the photoelectrons that have been created close to or at the surface) can escape undisturbed through the sample surface into the vacuum above the sample. The photoelectrons travel through the solid and quickly interact with the surrounding material, thereby loosing their initial kinetic energy. The X-rays excite electrons from the strongly bound core levels in the sample, creating photoions and photoelectrons.  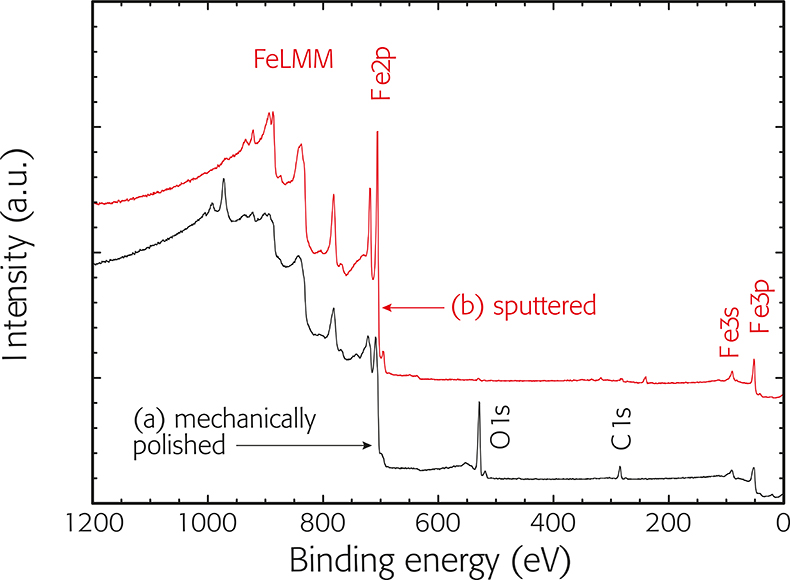
The samples are exposed to X-rays (typically AlKa or MgKa with 1486 eV and 1256 eV photon energy, respectively) under ultra-high vacuum (UHV) conditions. X-Ray photoelectron spectroscopy (XPS), also known as electron spectroscopy for chemical analysis (ESCA) is a surface sensitive spectroscopy that allows to determine the chemical composition of surface layers of solid samples.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
